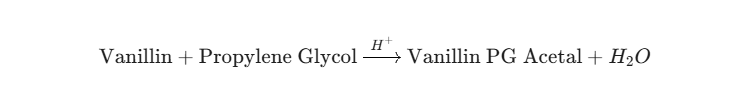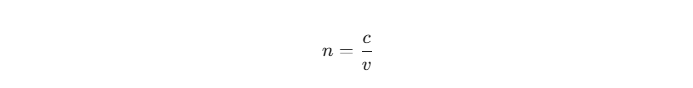Nostalgia Marketing: Why 90s Candy Flavors Are Booming in the E-Liquid Industry
Author: R&D Team, CUIGUAI Flavoring
Published by: Guangdong Unique Flavor Co., Ltd.
Last Updated: Apr 03, 2026

Candy vs. Chemistry
As flavor formulators and e-liquid manufacturers, we understand the immense pressure you face to create the next “All-Day Vape” (ADV) in a market that feels perpetually saturated. It is tempting to believe that adult consumers strictly desire sophisticated, mature flavor profiles—such as rich oak-barrel bourbons, complex botanical gins, or robusto tobaccos. However, market realities dictate a starkly different narrative. Today’s adult consumers are overwhelmingly reaching for the unapologetically sweet, neon-tinted, and hyper-sensory flavors of their childhood.
Welcome to the booming economy of 90s candy flavors.
Nostalgia marketing is far more than a retro aesthetic or a clever packaging gimmick; it is a deeply rooted psychological and chemical phenomenon. By tapping into “rewind culture,” brands are bypassing modern consumer fatigue and connecting directly with the brain’s emotional core. In this comprehensive technical guide, we will deconstruct the science of nostalgia, break down the chemical formulations behind iconic 90s confectionery profiles, and explore the unique engineering challenges of translating these retro flavors into premium e-liquids.
I、The Psychology of Nostalgia: Why “Rewind Culture” Drives Sales
To understand why a 35-year-old consumer prefers a “Blue Razz” disposable vape over a traditional tobacco blend, we must first look at the neuroscience of taste and memory.
The olfactory bulb, which processes aroma, has direct neural pathways to the amygdala and hippocampus—the regions of the brain responsible for emotion and episodic memory. This physiological wiring creates the “Proustian memory effect,” where a specific flavor or scent can instantly trigger a vivid, emotionally charged memory from the past.
For Millennials and older Generation Z cohorts, the 1990s and early 2000s represent a distinct era of sensory extremity. It was the golden age of hyper-sour gummies, color-changing lollipops, and blue-tinted beverages. In today’s fast-paced and often chaotic world, consumers experience a psychological phenomenon known as “rosy retrospection,” where the brain subconsciously idealizes the past.
The Behavioral Science Reality: According to research published in Frontiers in Psychology, nostalgia triggered by taste and scent stimuli bypasses standard cognitive purchasing filters. It directly facilitates deep emotional involvement and psychological comfort, which rapidly converts into consumer behavioral intention and brand loyalty.
When you market a “90s Sour Watermelon” e-liquid, you are not just selling a profile built on organic compounds; you are selling a 30-milliliter bottle of psychological comfort, simplicity, and joy.
II、The Anatomy of a 90s Candy Flavor: A Technical Deconstruction
What exactly defines a 90s candy flavor? Unlike modern artisanal profiles that strive for hyper-realistic, botanical accuracy, 90s flavors are intentionally exaggerated. They do not taste like fruit plucked from a tree; they taste like the idea of a fruit. They are characterized by extreme sweetness, sharp acidic kicks, and a distinct lack of earthy or fibrous base notes.
Below, our flavor chemists deconstruct the volatile organic compounds responsible for three of the most iconic retro profiles.
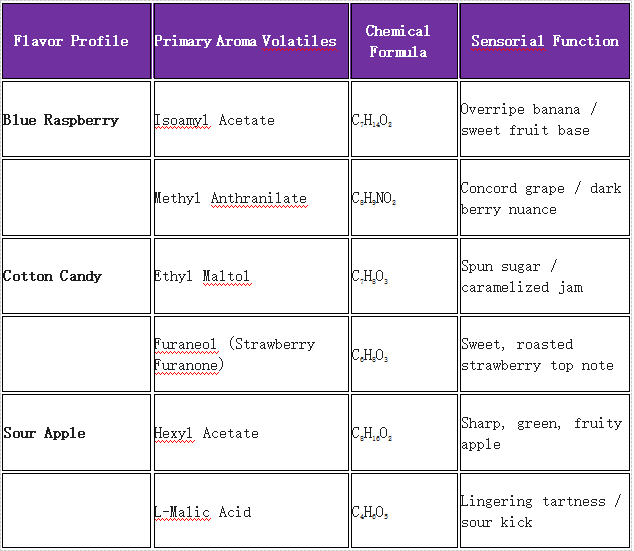
1. The Blue Raspberry Phenomenon
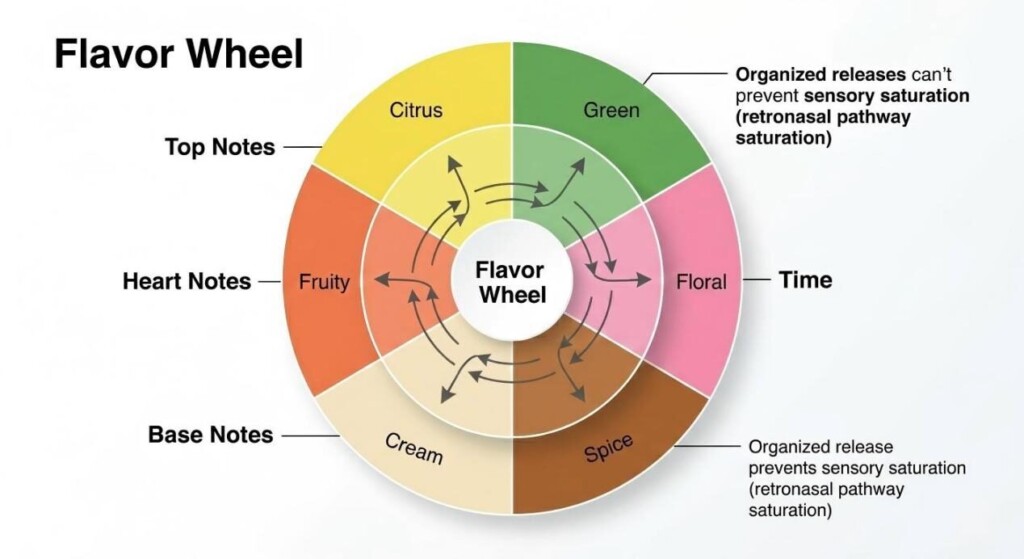
“Blue Raspberry” is arguably the crown jewel of the vaping industry, yet it does not exist in nature. While some point to the whitebark raspberry (Rubus leucodermis) as an inspiration, the modern “Blue Razz” profile is entirely a triumph of synthetic flavor design.
Historically, this flavor was born out of regulatory necessity. Following the FDA’s 1976 ban on the food dye FD&C Red No. 2 (Amaranth) due to safety concerns, confectioners needed a new color to represent raspberry and differentiate it from cherry and strawberry. They pivoted to Brilliant Blue FCF, and thus, the “Blue Raspberry” standard was cemented in the consumer psyche.
Chemically, Blue Razz is a complex fantasy fruit built on a foundation of specific esters and aldehydes:
- Isoamyl Acetate (C7H14O2):Provides an overripe, candy-like banana nuance that rounds out the sharp berry notes.
- Methyl Anthranilate (C8H9NO2):Delivers a deep, sweet, Concord grape characteristic that adds body.
- Allyl Hexanoate (C9H16O2):Contributes a sharp, syrupy pineapple top note.
When expertly blended, these compounds create a jammy, tart, and multi-layered profile that registers to the brain instantly as “Blue Razz.”
Molecular Flavor
2. Spun Sugar and Cotton Candy
Cotton candy is the epitome of the fairground experience, relying on an olfactory profile that smells like heat, caramelized sugar, and pure sweetness. In flavor chemistry, the backbone of cotton candy is Ethyl Maltol (C7H8O3).
Ethyl maltol is a synthetic organic compound that acts as a powerful flavor enhancer. On its own, it possesses a distinct aroma of cooked fruit and caramelized sugar. To build a true 90s pink cotton candy e-liquid, formulators typically combine:
- Ethyl Maltol (C7H8O3):The primary spun-sugar base.
- Furaneol (C6H8O3):Also known as strawberry furanone, this adds the subtle, roasted, jammy-strawberry top note characteristic of “pink” cotton candy.
- Vanillin (C8H8O3):Provides a creamy, marshmallow-like backend to smooth the profile.
3. Extreme Sour Apple and Watermelon
The 90s candy scene was defined by the “extreme sour” craze. Hard candies and gummies coated in malic acid dust dominated the market.
- Sour Apple:Built around Hexyl Acetate (C8H16O2), which delivers a sharp, green, almost solvent-like fruity apple note, completely devoid of the mellow oxidation found in real apples.
- Candy Watermelon:Relies heavily on Melonal (C9H16O), an aldehyde that offers a powerful, penetrating melon aroma, paired with Ethyl Methylphenylglycidate (the classic “strawberry aldehyde”) to push it into the realm of candy rather than fresh fruit.
4. Chemical Profile Quick Reference
III、Translating Confectionery to E-Liquids: Formulation Challenges
While it is easy to identify the aroma volatiles used in a gummy worm, translating those compounds into a combustible or vaporized format is a complex engineering challenge. E-liquid formulators face thermodynamic hurdles that food chemists simply do not. We cannot just dump confectionary flavorings into a Propylene Glycol (PG) and Vegetable Glycerin (VG) base and expect a premium vaping experience.
1.Heat Degradation and Coil Gunking
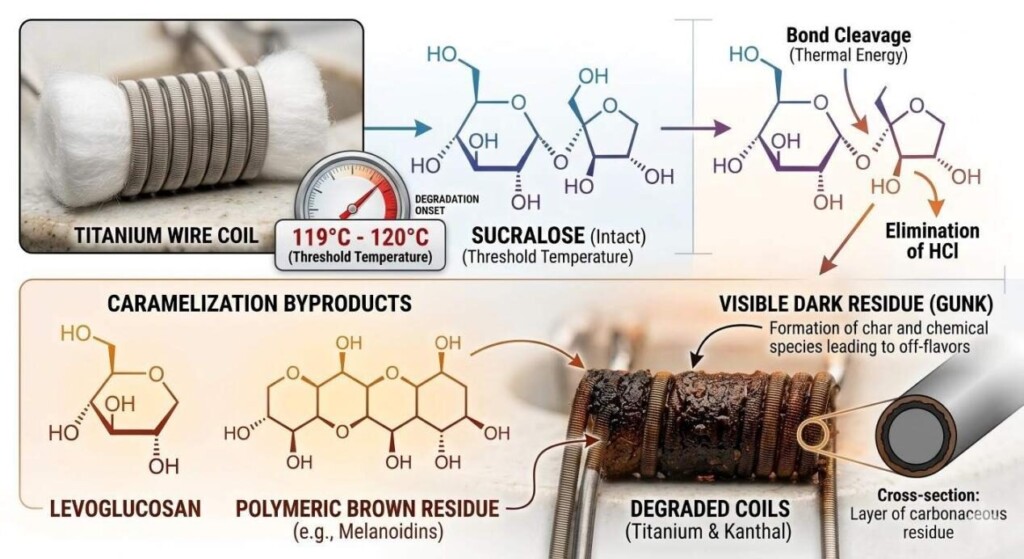
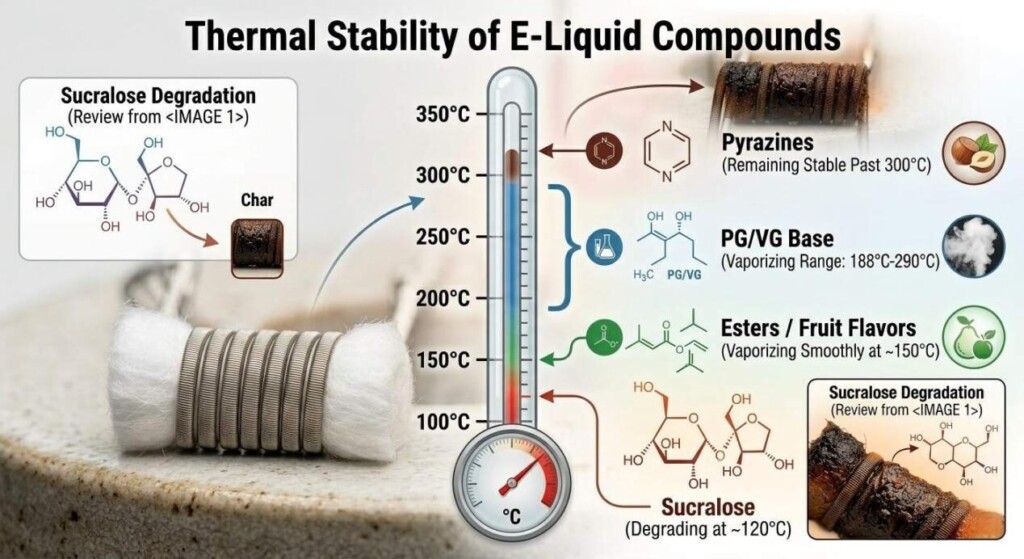
In traditional food manufacturing, sweeteners like sucrose and corn syrup are standard. In e-liquids, these sugars are strictly forbidden as they combust, releasing toxic byproducts and immediately carbonizing on the heating element (a phenomenon known in the industry as “coil gunk”).
Instead, e-liquid manufacturers rely on high-intensity artificial sweeteners like Sucralose or natural compounds like Stevia. However, even these must be carefully calibrated. Furthermore, components like Ethyl Maltol have strict solubility limits. Ethyl maltol is only soluble in Propylene Glycol up to approximately 10% at room temperature. If a formulator attempts to push the concentration higher to achieve a sweeter “cotton candy” note, the compound will recrystallize and crash out of the solution, rendering the e-liquid unusable and destroying the user’s vape hardware.
2.Balancing the “Sour Kick”
Replicating the intense sourness of 90s candy is perhaps the most difficult technical hurdle. In food, formulators use high concentrations of Citric Acid, Tartaric Acid, or L-Malic Acid. However, when vaporized, heavy organic acids do not aerosolize cleanly. Over-acidification of an e-liquid will lead to rapid degradation of the metal coil, muting the flavor after just a few milliliters of use.
To achieve a sustainable “sour” profile, elite flavor chemists use trace amounts of Malic Acid purely to pull the pH down slightly, and simulate the sensation of sourness by utilizing sharp, volatile citrus aldehydes (like Citral) combined with synthetic cooling agents.
3.The Role of Cooling Agents (WS-23)
Modern disposable vapes have perfected the candy profile by pairing it with synthetic cooling agents, most notably WS-23 (N,2,3-trimethyl-2-isopropylbutanamide). Unlike traditional menthol, which carries a distinct minty, earthy flavor that can clash with sweet candy profiles, WS-23 provides a pure, clean thermal cooling sensation to the back of the throat without altering the primary flavor. This coldness tricks the palate into perceiving the candy flavors as “fresher” and sharper, perfectly complementing the heavy sweetness of a Blue Razz or Watermelon profile.

The Mesh Coil
IV、Market Data: The Economic Boom of Nostalgia Flavors
If the chemistry hasn’t convinced you of the viability of 90s candy flavors, the market data certainly will. The e-cigarette industry has experienced a massive paradigm shift, moving aggressively away from traditional tobacco analogs and leaning heavily into the confectionary space.
The Market Reality: Data tracked by the CDC Foundation’s Monitoring Tobacco Product Use report highlights a staggering shift in consumer behavior. As of late 2025, non-tobacco-flavored e-cigarette sales (specifically fruit, mint, candy, and cooling blends) accounted for nearly 79% of total unit sales in the US market. Furthermore, within the disposable vape sector, that number skyrockets to over 91%.
Similarly, a comprehensive market analysis published in the journal Tobacco Control (BMJ) reviewed tens of thousands of e-liquids across European markets. The study revealed that sweet profiles—particularly categorized under candy, dessert, and fruit—absolutely dominate the global market. The sheer universal palatability of these sweet descriptors transcends age demographics, proving that adult consumers are the primary drivers of the candy-flavor boom.
For an e-liquid brand, ignoring the data and failing to offer high-quality, nostalgic candy profiles is equivalent to leaving substantial revenue on the table.
V、Regulatory Compliance and Safety in Flavor Manufacturing
While chasing the 90s flavor trend is highly lucrative, it must be done with an uncompromising commitment to safety and regulatory compliance. The landscape of inhalation toxicology is vastly different from gastrointestinal safety. An ingredient that is Generally Recognized As Safe (GRAS) for ingestion by the Flavor and Extract Manufacturers Association (FEMA) is not automatically safe for inhalation.
As a reputable manufacturer, we ensure that our nostalgic flavor profiles are engineered specifically for the e-liquid sector:
- Diacetyl and Acetyl Propionyl Free:The creamy, buttery notes sometimes used to round out candy profiles must be completely devoid of Diacetyl and Acetyl Propionyl to prevent severe respiratory risks (such as bronchiolitis obliterans).
- TPD and PMTA Readiness:Whether you are formulating for the European Union’s Tobacco Products Directive (TPD) or navigating the FDA’s Premarket Tobacco Product Application (PMTA) pathway in the United States, your raw flavorings must come with complete chemical transparency, Gas Chromatography-Mass Spectrometry (GC-MS) reporting, and toxicological risk assessments.
We do not just provide a “cotton candy” flavoring; we provide the exact molecular breakdown and safety data sheets required to keep your brand on the shelves legally.
VI、The Future of E-Liquid Flavoring: Beyond the 90s

Vape Innovation
The nostalgia marketing boom is not a fleeting trend; it is a foundational pillar of the modern vaping industry. However, consumer palates will continue to evolve. The next wave of innovation will not just be about recreating the 90s, but elevating it.
We are already seeing the emergence of “hybrid nostalgia”—classic 90s candy bases engineered with exotic, modern twists. Think Blue Raspberry Dragonfruit, or Sour Yuzu Watermelon. By utilizing advanced extraction techniques and ultra-pure synthetic isolates, flavor manufacturers can create profiles that offer the psychological comfort of the past with the sophisticated complexity of the future.
The brands that will dominate the market for the next decade are the ones that understand the delicate balance between emotional marketing and rigorous flavor science.
VII、Conclusion & Next Steps
Formulating a successful e-liquid in today’s highly competitive landscape requires more than just mixing off-the-shelf ingredients. It requires an understanding of consumer psychology, market economics, and uncompromising chemical engineering. Nostalgia marketing, driven by the timeless appeal of 90s candy flavors, offers a proven, highly profitable pathway for brands willing to invest in premium formulation.
Do not let your product line fall behind the curve. Whether you need to reformulate an existing profile to prevent coil degradation, or you are looking to launch a brand-new line of compliance-ready retro candy vapes, our team of expert flavor chemists is ready to assist.
Ready to engineer your next best-seller? Partner with us to bring the ultimate 90s nostalgia profiles to your product line safely, legally, and deliciously.
Let’s Talk Formulation:
Technical Exchange & Free Samples: Schedule a consultation with our master flavorists and request your custom sample pack today.
| Contact Channel | Details |
| 🌐 Website: | www.cuiguai.com |
| 📧 Email: | info@cuiguai.com |
| ☎ Phone: | +86 0769 8838 0789 |
| 📱 WhatsApp: | +86 189 2926 7983 |
| 📍 Factory Address | Room 701, Building 3, No. 16, Binzhong South Road, Daojiao Town, Dongguan City, Guangdong Province, China |