Author:R&D Team, CUIGUAI Flavoring
Published by:Guangdong Unique Flavor Co., Ltd.
Last Updated:Nov 15, 2025

Modern Aroma Lab & Formulation
In the competitive world ofelectronic liquid (e-liquid) flavor manufacturing, success depends on how fast a company can create and validate new flavors—without compromising stability, safety, or sensory quality. Consumers demand novelty: tropical fruit explosions, creamy dessert bases, and authentic tobacco blends. Yet, every innovation must also meet the highest standards ofchemical consistency, thermal stability, eregulatory compliance.
For a manufacturer, developing100 stable flavorsis not just an achievement—it’s anengineering systemthat blends chemistry, sensory science, and quality management. The process involves data-driven formulation, raw material traceability, accelerated stability testing, and structured creativity.
This article provides atechnical roadmapfor how leading flavor manufacturers likeAromatizante CUIGUAIcan efficiently develop and stabilize 100+ e-liquid flavors while maintaining precision, reproducibility, and compliance across global markets.
Flavor stability refers tothe ability of a flavor formulation to maintain its sensory and chemical integrityunder normal storage, transportation, and usage conditions. In scientific terms, it involves controlling:
A stable flavor maintains itsprofile, strength, and clarityover its intended shelf life, typically12–24 months.
For consumers, stability equals trust. An unstable flavor may darken, lose aroma intensity, or even develop unwanted notes after just weeks of storage. This can lead to:
According tothe Flavor and Extract Manufacturers Association (FEMA), stability and safety are foundational to ensuring consumer confidence in flavor products (FEMA, 2023).
To develop 100 stable flavors quickly, a manufacturer must balancespeed and science. Modern flavor houses usedata-driven formulation pipelines—where analytical chemistry, sensory evaluation, and AI-based prediction models guide rapid iteration.
Let’s explore the pillars of this approach.
A successful flavor program starts with aningredient intelligence database. This database catalogs:
Such databases allow formulators topredict interactionsand design stable blends faster. Many manufacturers integrate this data withGC–MS reference chromatogramseinternal sensory panels.
Rather than starting each flavor from scratch, leading companies use amodular design approach:
This modular system enables the creation of 100+ distinct flavors throughstructured recombination, reducing development time by over 60%.
According toa 2022 study in the Journal of Food Science, modular aroma design and pre-characterized ingredient databases significantly improve flavor reproducibility and shorten R&D timelines (Journal of Food Science, 2022).
Every new flavor is analyzed usingGas Chromatography–Mass Spectrometry (GC–MS)to ensure purity and detect potential reactive compounds. This analytical fingerprint helps predict:
The combination ofGC–MS + sensory evaluationforms the backbone ofrapid yet controlled innovation.
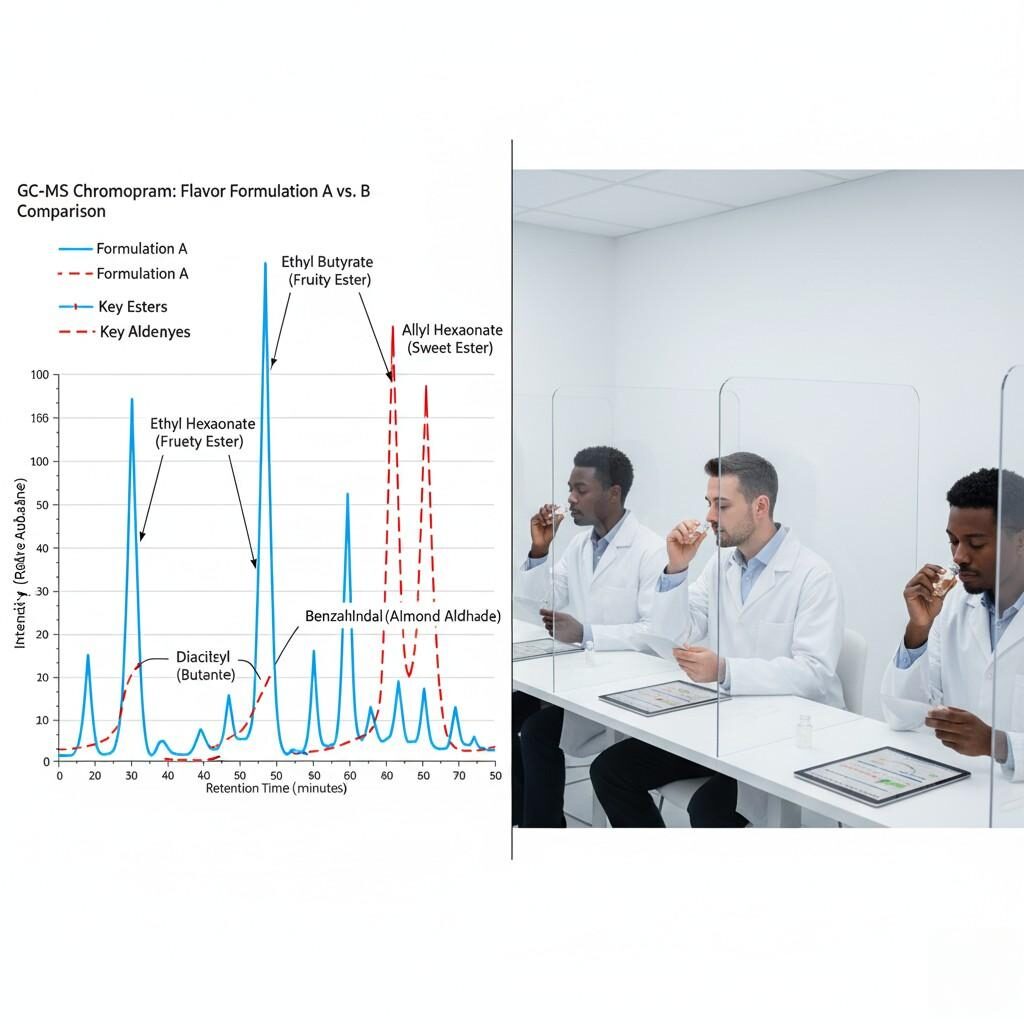
E-Liquid Flavor GC-MS & Sensory
Oxidation is the primary cause of flavor degradation. Compounds like vanillin, benzaldehyde, or limonene oxidize easily, producing off-notes or color shifts.
To control oxidation:
OU.S. National Library of Medicinenotes that oxygen exposure accelerates the breakdown of volatile organic molecules in food and fragrance systems, especially under light and heat (PubChem, 2024).
The carrier system—usuallyPropileno glicol (PG)eGlicerina vegetal (VG)—influences solubility and volatility.
Solvent polarity and viscosity directly affectmolecular dispersion, flavor strength, estorage stability.
Some flavor molecules (like vanillin or citral) are sensitive to pH shifts and react with nicotine bases or acids. To prevent unwanted reactions:
High-speed blending without temperature or oxygen control can damage fragile compounds. Modern flavor factories use:
CUIGUAI Flavoring employs such controlled systems to preserve volatile esters and maintain aroma precision.
To certify a flavor as “stable,” it must undergoaccelerated stability testing—simulating months of storage in just weeks. Tests typically involve:
Data is recorded usingchromatographic peak area analysiseorganoleptic scoring.
| Parâmetro | Método | Propósito |
| Peroxide Value | Titration | Detects oxidation progress |
| GC–MS fingerprint | Analytical chemistry | Monitors compound stability |
| pH drift | Electrometric | Identifies acid/base instability |
| Sensory evaluation | Panel testing | Tracks aroma consistency |
These results are compiled into aStability Index, used to classify flavors into categories:
Machine learning models trained onhistorical stability datacan predict which new formulations will likely remain stable.
Using such predictive analytics can reducereal-world testing time by up to 50%, accelerating flavor development from months to weeks.
According toMIT’s Chemical Engineering Department, predictive modeling and machine learning are transforming formulation science across food, cosmetic, and vaping industries by enabling faster, data-backed innovation (MIT OpenCourseWare, 2023).

E-Liquid Stability Testing
To create100 stable flavors efficiently, companies need an organized workflow combiningchemistry, sensory, and production engineering.
A digitalProduct Lifecycle Management (PLM)system ensures traceability across all stages.
Dividing R&D teams intothematic clusters(e.g., Fruit, Dessert, Beverage, Tobacco, Mint) allows parallel progress. Each team maintains its ownmini flavor library, contributing to the 100-flavor target collaboratively.
Each stable flavor must be accompanied by:
Having these ready shortens the PMTA/TPD submission process later.
Impure ingredients introduce instability. Collaborate only with suppliers offering:
CUIGUAI Flavoring employs athree-tier supplier auditsystem:
This ensuresraw material uniformity, preventing variability across hundreds of flavor formulations.
Scaling up a stable prototype to full production requires careful process control.
Each flavor batch must follow identical:
Automated PLC systems ensure reproducibility, minimizing human error.
Containers must prevent oxygen, UV, and moisture ingress. Amber glass or coated PET bottles are standard for sensitive flavors.
CUIGUAI Flavoring also usesinert gas fillingevacuum sealingfor long-term stability during export.
With 100+ flavors, data management becomes a core challenge.
Acentralized digital platformcan manage:
Such systems simplifyregulatory reportingeclient communication, especially for PMTA or EU-TPD submissions.
Even the most advanced analytical data must align withhuman sensory evaluation. CUIGUAI Flavoring integrates:
Consistency betweeninstrumental data and human perceptiondefines final product success.
Fast development often risks instability, but modern science allows both speed and stability.
The key is buildingpredictive controlat every step—from ingredient selection to final shelf testing.
At CUIGUAI Flavoring, we see the creation of 100 stable flavors not as mass production but as100 parallel scientific achievements, each rooted in chemistry, technology, and sensory design.

Flavor Development & Evaluation Collage
The ability to develop 100 stable flavors rapidly is not luck—it isthe result of scientific discipline, data intelligence, and manufacturing excellence.
By integratingchemical analytics, predictive modeling, GMP production, and sensory expertise, a manufacturer can innovate faster while ensuring every flavor meets global safety and quality expectations.
CUIGUAI Flavoring stands at the intersection ofcreativity and compliance, offering:
Fortechnical collaboration, PMTA documentation supportoufree sample requests, contact our technical team today:
📧 Email: [info@cuiguai.com]
🌐 Website: [www.cuiguai.com]
📱 WhatsApp: [+86 189 2926 7983]
☎ Phone: [+86 0769 8838 0789]
Let’s build the next generation ofstable, compliant, and innovative flavorstogether.
O escopo de negócios inclui projetos licenciados: produção de aditivos alimentares. Projetos gerais: vendas de aditivos alimentares; fabricação de produtos químicos diários; vendas de produtos químicos diários; serviços técnicos, desenvolvimento de tecnologia, consultoria técnica, intercâmbio de tecnologia, transferência de tecnologia e promoção de tecnologia; pesquisa e desenvolvimento de rações biológicas; pesquisa e desenvolvimento de preparação enzimática industrial; atacado de cosméticos; agência de comércio doméstico; vendas de produtos sanitários e suprimentos médicos descartáveis; varejo de utensílios de cozinha, louças sanitárias e artigos diversos; vendas de necessidades diárias; vendas de alimentos (apenas vendas de alimentos pré-embalados).
Copyright ©Guangdong Unique Flavor Co., Ltd.All Rights Reserved. Privacy Policy Return and Exchange Policy