Author:R&D Team, CUIGUAI Flavoring
Published by:Guangdong Unique Flavor Co., Ltd.
Last Updated: Dec 04, 2025
Blockchain Supply Chain Traceability
The transition of the electronic liquid industry from a niche market to a globally scrutinized sector has introduced a fundamental requirement for all manufacturers:Flavor Transparency. Under the relentless pressure of multinational regulations, including the U.S. Food and Drug Administration’s (FDA) Premarket Tobacco Product Application (PMTA) pathway and the European Union’s Tobacco Products Directive (TPD), opacity is no longer tenable. For flavor manufacturers, achieving transparency is not merely a compliance task; it is the ultimate expression of quality, scientific rigor, and a critical determinant of market access.
This comprehensive technical guide is dedicated to outlining the architecture of flavor transparency in the modern era. We explore the regulatory mandates, the scientific standards required to meet them, and the systematic manufacturing practices that transform intellectual property (IP) into verifiable, consumer-safe data.
Flavor transparency in e-liquids is driven by three interconnected regulatory demands:Toxicity, Traceability, eConsistency. Unlike the food industry, where flavor disclosure is often governed by simple labeling laws, the e-liquid industry requires granular, highly specific chemical data for every ingredient that will be aerosolized and inhaled.
The core of multinational regulation requires manufacturers to prove that their product is"Apropriado para a proteção da saúde pública".This burden of proof directly translates into an absolute demand for flavor transparency.
This shift means the traditional practice of labeling flavors as merely “Artificial Flavoring” or “Natural Flavoring” is inadequate for regulatory submission. Transparency must be provided in the form of aComplete Ingredient ManifesteToxicological Dossierfor every flavor concentrate sold to a client.
OFlavor and Extract Manufacturers Association (FEMA)GRAS (Generally Recognized As Safe) program is the global standard for flavor safety. However, the regulatory foundation of transparency begins with a critical distinction that FEMA itself asserts:GRAS status applies to ingestion, not inhalation.
FEMA’s robust assessment process, which is often cited as the gold standard in ingredient safety, does not specifically evaluate flavor ingredients for use in e-cigarettes. This creates a secondary responsibility for the flavor manufacturer:
This dual validation process—GRAS for food, safety data for aerosolization—is a cornerstone of flavor transparency under multinational regulations.
Building a transparent flavor system requires embedding quality and disclosure into the physical and procedural foundation of the manufacturing process. Certifications and methodologies serve as the external verification of a transparent internal system.
OInternational Organization for Standardization (ISO)eGood Manufacturing Practices (GMP)are not regulatory bodies in the same way as the FDA, but they provide the auditable framework essential for transparency.
Manufacturers who adhere to these standards build trust by demonstrating that their transparency claims are supported by a globally recognized, independently audited system.
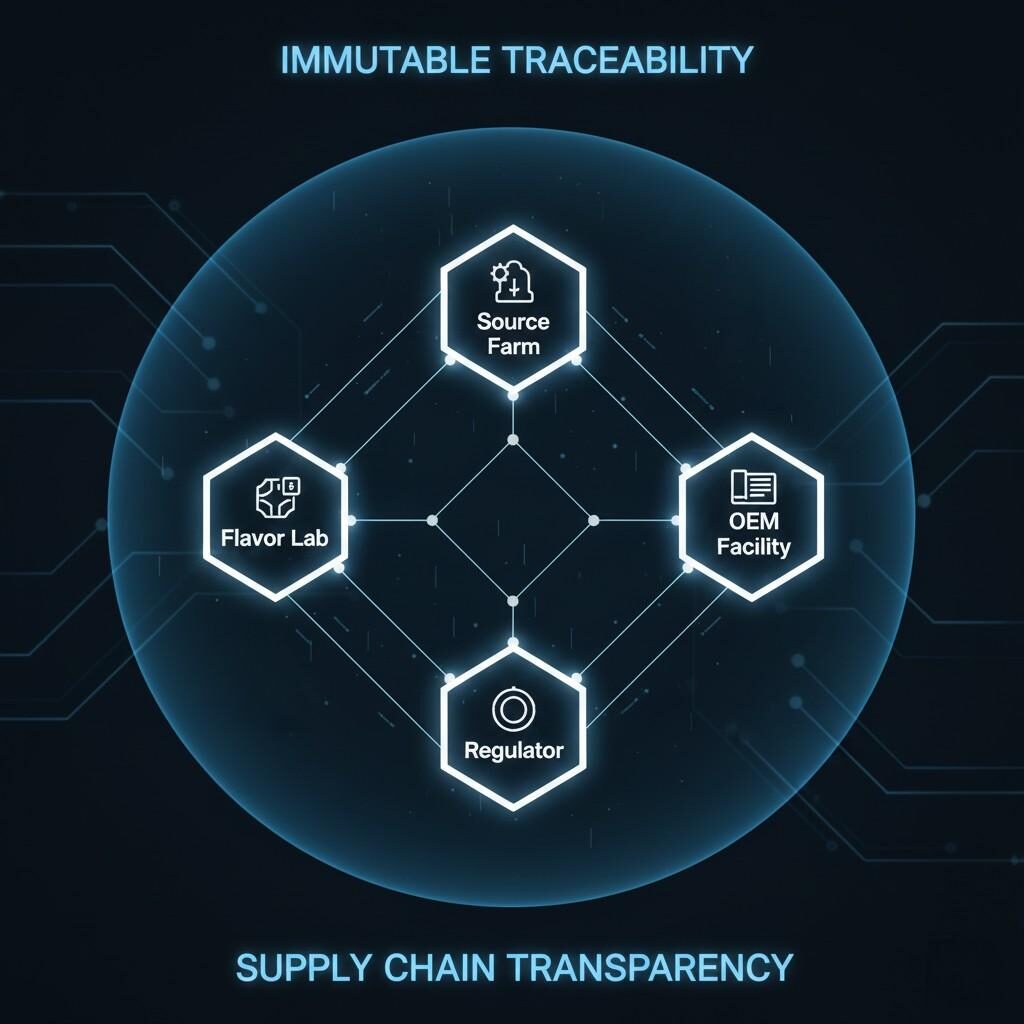
Traceability is the operational engine of transparency. Regulatory submissions require not justwhatis in the flavor, butwhereit came from andhowit was handled. This necessitates a three-tiered approach to supply chain disclosure:
Advanced technologies, such asBlockchain and RFID tracking, are increasingly being adopted to create an immutable record of ingredients across complex, multi-tiered global supply chains, further solidifying the commitment to verifiable transparency.

Regulatory Compliance Dashboard
Transparency is ultimately validated through scientific data. The flavor industry must utilize advanced analytical tools to prove that the ingredient data submitted to regulators is both accurate and consistent.
The workhorse of flavor transparency isGas Chromatography-Mass Spectrometry (GC-MS). This technique separates the flavor concentrate into its individual components and quantifies the amount of each compound present.
Since the e-liquid is heated, transparency demands proof of what happens when the flavor ingredients are subjected to thermal stress.Pyrolysis studiessimulate the coil heating process.
This is the ultimate test of transparency: showing the regulator not just what the consumerbuys, but what the consumerinhales.

E-Cigarette Regulatory Testing
For the flavor manufacturer, the investment in transparency creates a powerful, defensible competitive advantage, fundamentally changing the B2B relationship with e-liquid brands.
A truly transparent flavor house provides clients with ready-made compliance documentation, transforming months of regulatory paperwork into a simple, traceable data transfer. This includes:
This service drastically reduces the regulatory burden and cost for the client, making the transparent flavor house an invaluable partner.
Multinational regulations are not static; they are tightening. Governments worldwide are increasingly using flavor restrictions (bans on certain flavorcategories) as a public health tool, as evidenced by flavor bans across various U.S. states (e.g., Massachusetts, New York) and countries (e.g., the Netherlands).
The transparent flavor manufacturer is best positioned to navigate these shifts because they possess granular data. They can rapidly reformulate a complex profile to remove a single newly restricted component, whereas an opaque recipe would require a complete, costly, and time-consuming re-engineering process.Data enables agility.
Transparency is moving beyond the regulator and into the consumer marketplace. Brands that provide QR codes on their packaging that link to a third-party verified Ingredient Authenticity Report are winning consumer trust. This level of retail transparency is only possible when the flavor manufacturer provides the upstream, verifiable data to support the final product claims.
The era of “secret recipes” in the e-liquid flavor industry is over. The rise of multinational regulations—from the precise reporting demands of the FDA and TPD to the foundational scientific skepticism of the FEMA GRAS program concerning inhalation—has madeFlavor Transparencythe central requirement for global market access.
For the flavor manufacturer, this shift demands investment in ISO/GMP systems, advanced analytical tools like GC-MS, and rigorous thermal testing protocols. By delivering not just a flavor profile, but a comprehensive, scientifically validatedTransparency Dossier, we enable our clients to achieve compliance, mitigate risk, and build the consumer trust necessary to succeed in a heavily scrutinized market.
The future belongs to the flavors that can prove their pedigree.

E-Liquid Regulatory Dossier
Is your product portfolio prepared for the next wave of multinational regulation?
We specialize in engineering flavor systems that meet the most stringent global transparency requirements. Contact us today to receive aComplimentary Technical Compliance Roadmapand access our library of PMTA/TPD-ready flavor concentrates.
📧 Email: [info@cuiguai.com]
🌐 Website: [www.cuiguai.com]
📱 WhatsApp: [+86 189 2926 7983]
☎ Phone: [+86 0769 8838 0789]
Partner with us to transform regulatory risk into market advantage.
O escopo de negócios inclui projetos licenciados: produção de aditivos alimentares. Projetos gerais: vendas de aditivos alimentares; fabricação de produtos químicos diários; vendas de produtos químicos diários; serviços técnicos, desenvolvimento de tecnologia, consultoria técnica, intercâmbio de tecnologia, transferência de tecnologia e promoção de tecnologia; pesquisa e desenvolvimento de rações biológicas; pesquisa e desenvolvimento de preparação enzimática industrial; atacado de cosméticos; agência de comércio doméstico; vendas de produtos sanitários e suprimentos médicos descartáveis; varejo de utensílios de cozinha, louças sanitárias e artigos diversos; vendas de necessidades diárias; vendas de alimentos (apenas vendas de alimentos pré-embalados).
Copyright ©Guangdong Unique Flavor Co., Ltd.All Rights Reserved. Privacy Policy Return and Exchange Policy